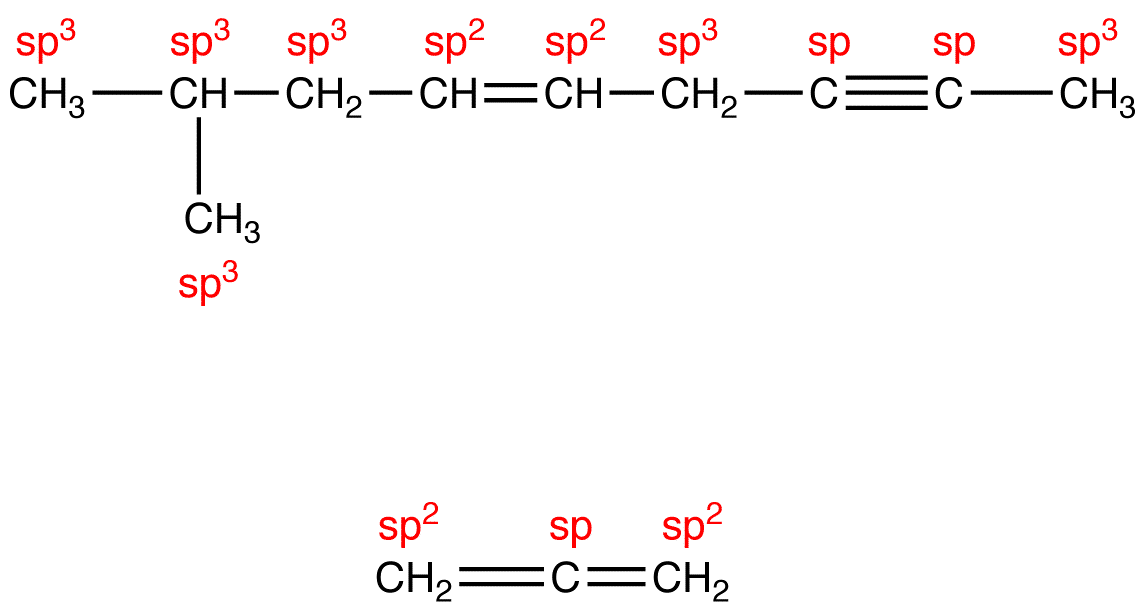
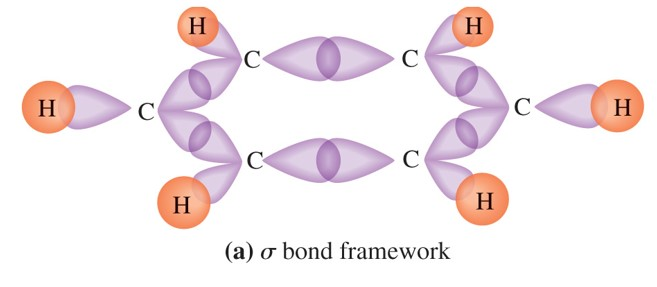
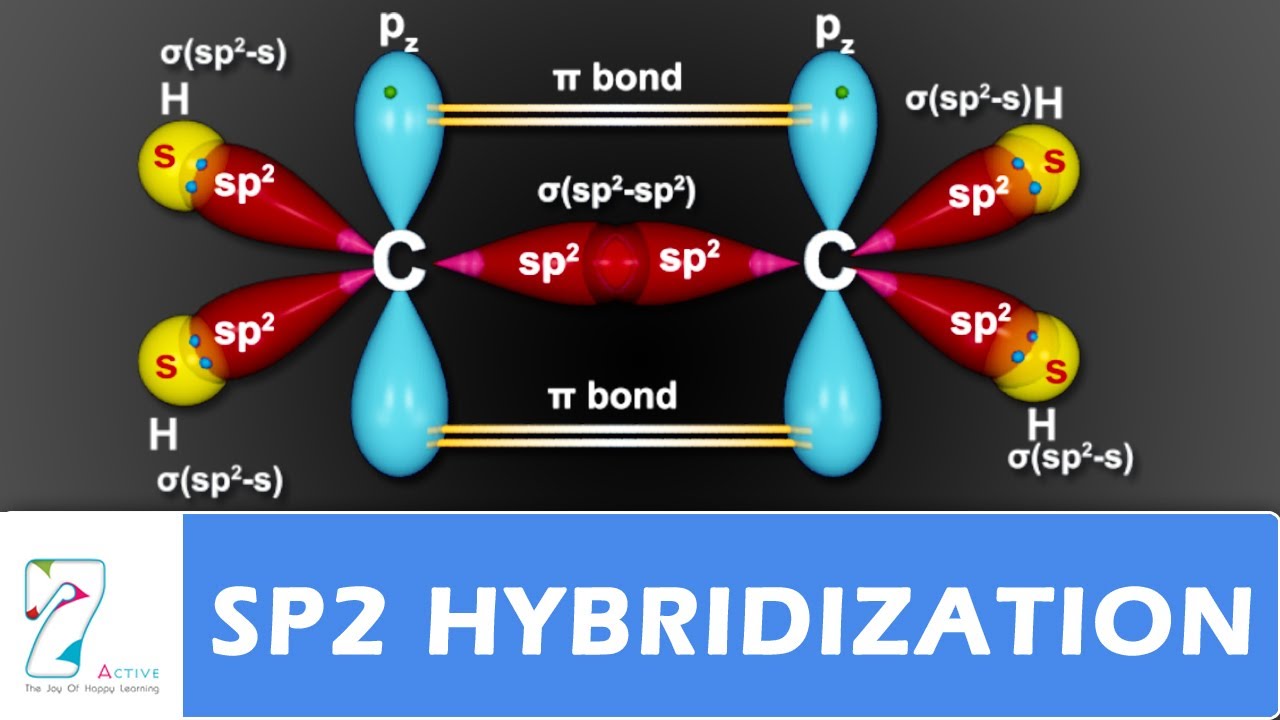
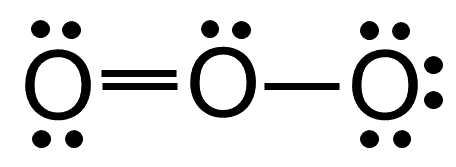Sp an example of this is acetylene (c 2 h 2 ).Count the number of lone pairs + the number of atoms that are directly attached to the central atom.
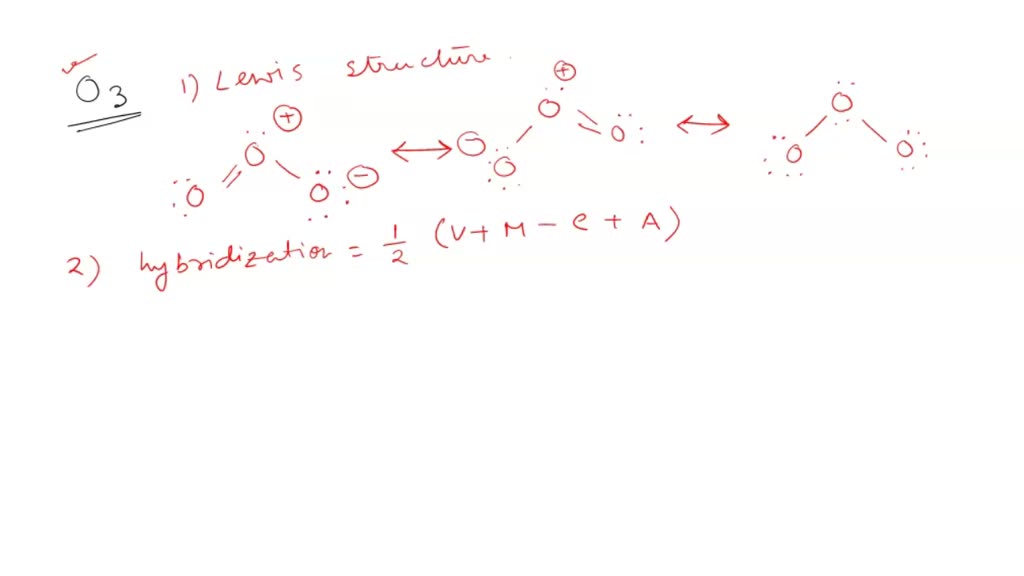
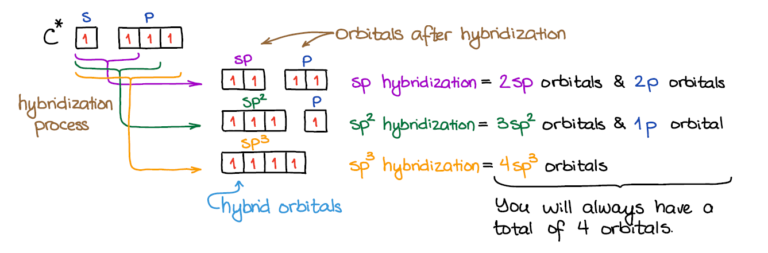
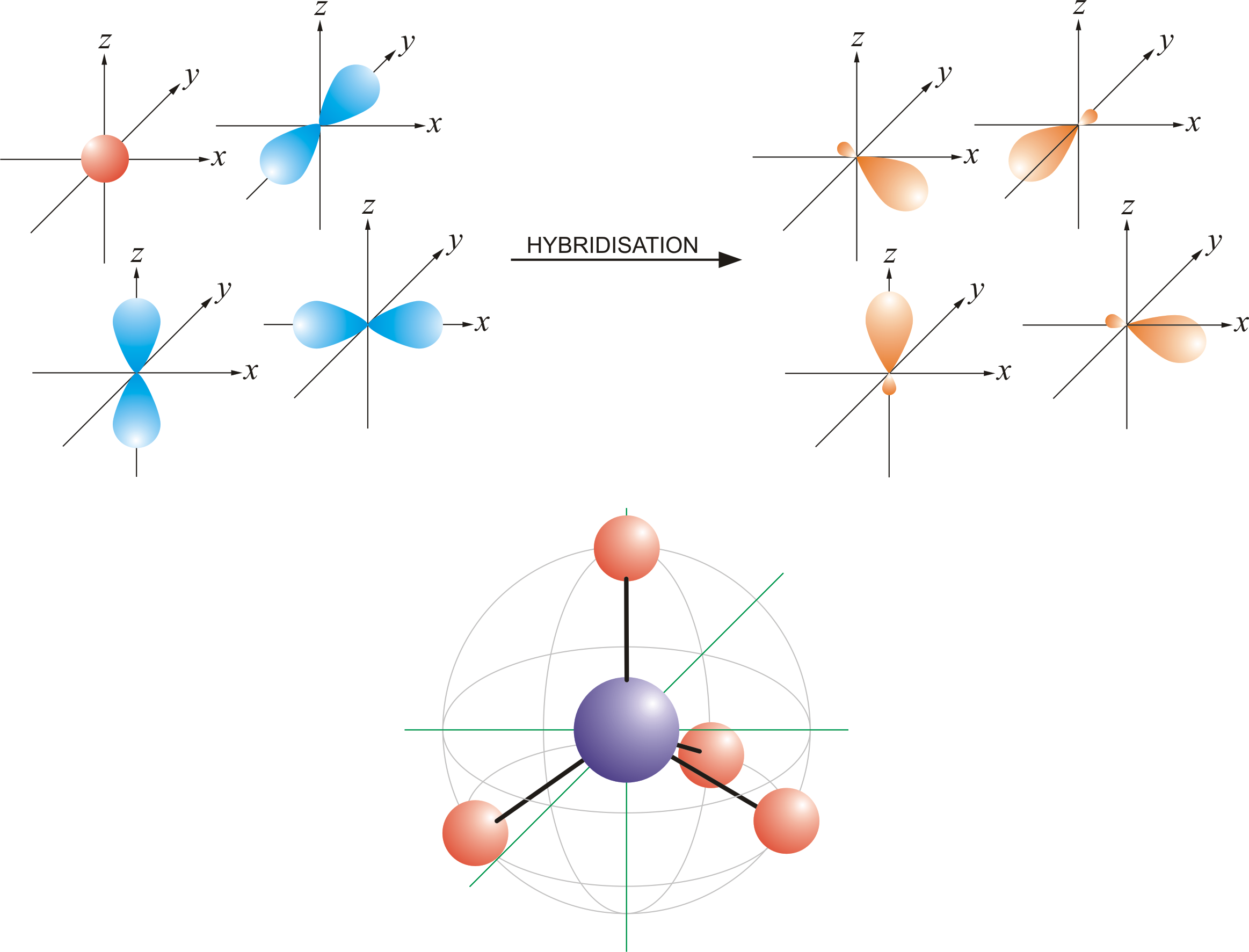
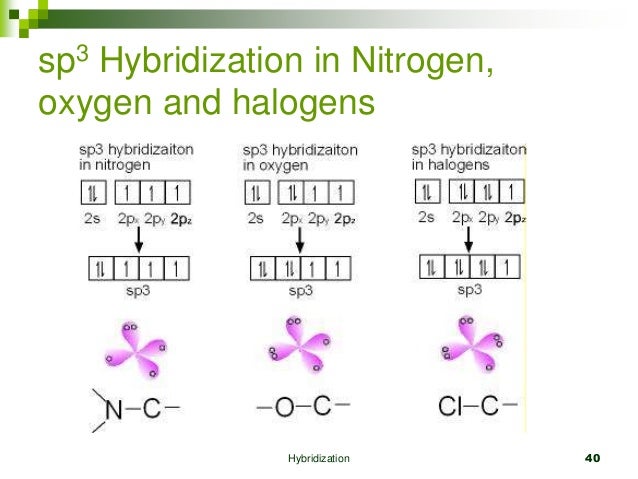
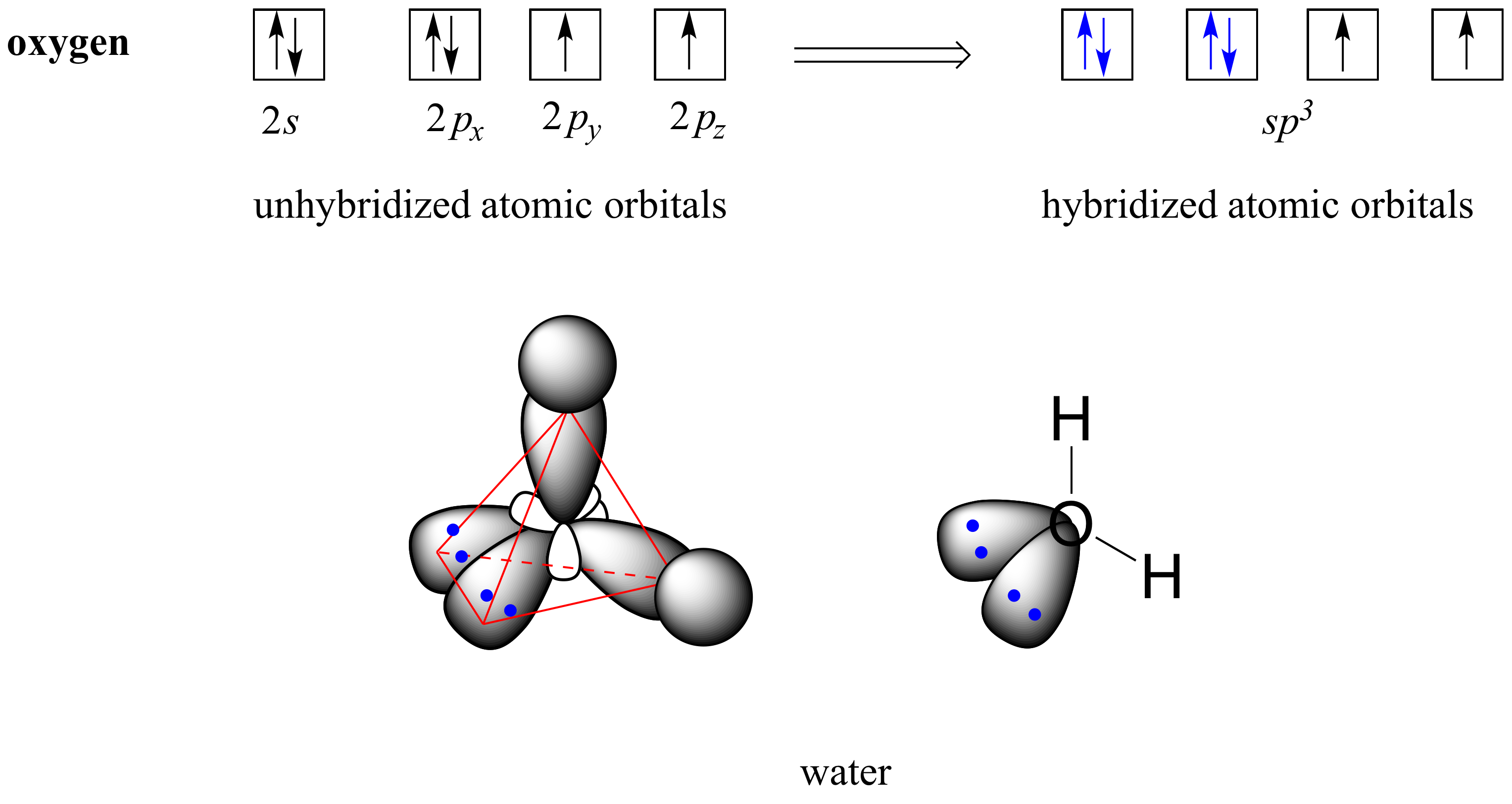
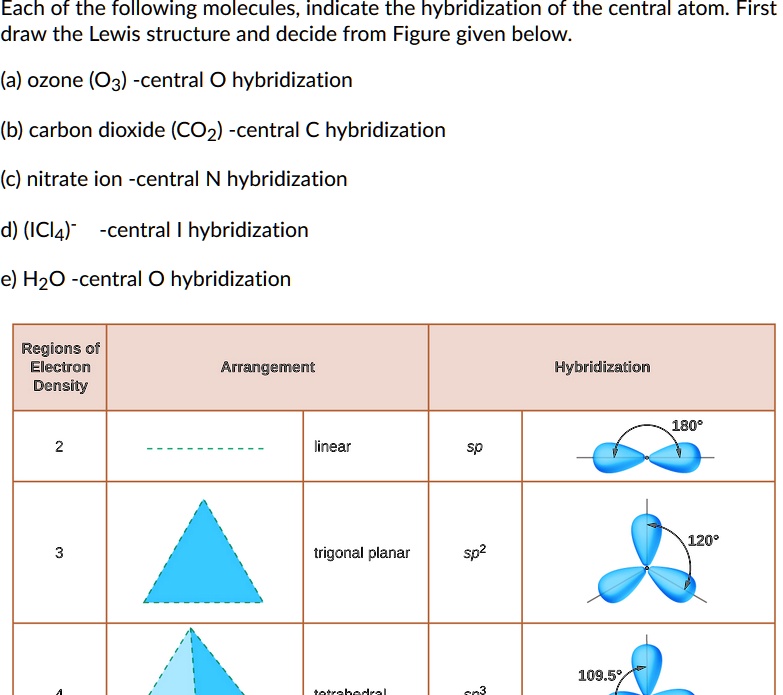
If it's 4, your atom is sp3.This chemistry video tutorial explains how to draw the lewis structure of o3.When one s and three p orbitals mix, a set of four equal hybrid orbitals, called sp 3 orbitals, is formed as illustrated in 1.3.2 1.3.
It also discusses the molecular geometry, bond angle, hybridization, and forma.This gives us equation 7.1.2, where the value 1 √2 is needed mathematically to indicate that the 2 s and 2 p orbitals contribute equally to each hybrid orbital.
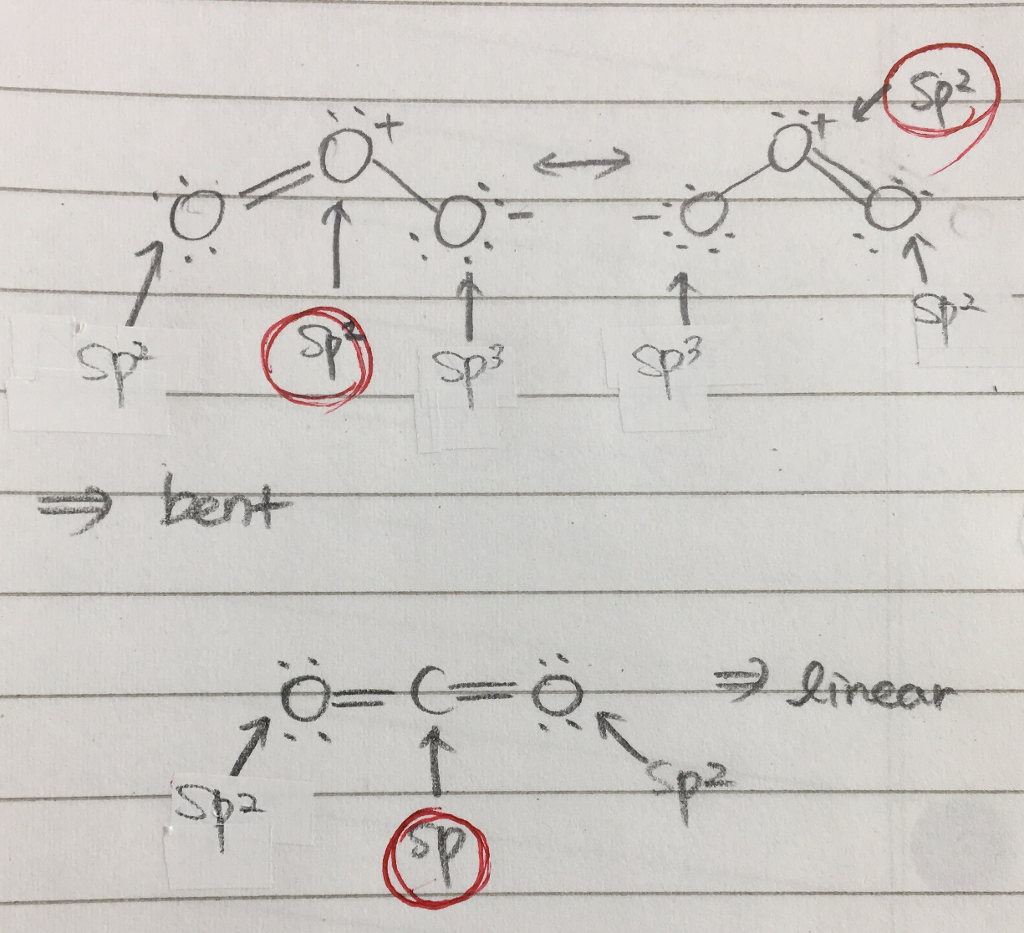
Carbon dioxide, co 2 , has a linear shape.To accommodate these two electron domains, two of the be atom's four valence orbitals will mix to.Figure 1a depicts an illustrative perovskite crystal structure and relative positions of the atoms in a lamo 3.the excitation and radiative decay channels are shown in fig.
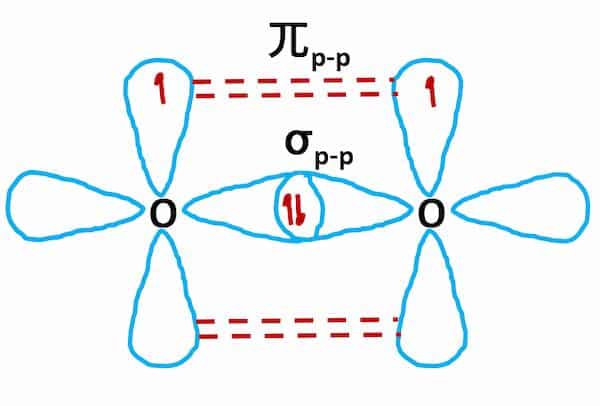
It comprises three oxygen atoms, out of which one oxygen atom is in the center forming a double bond.Graphene is composed of a uniform monolayer of carbon atoms.
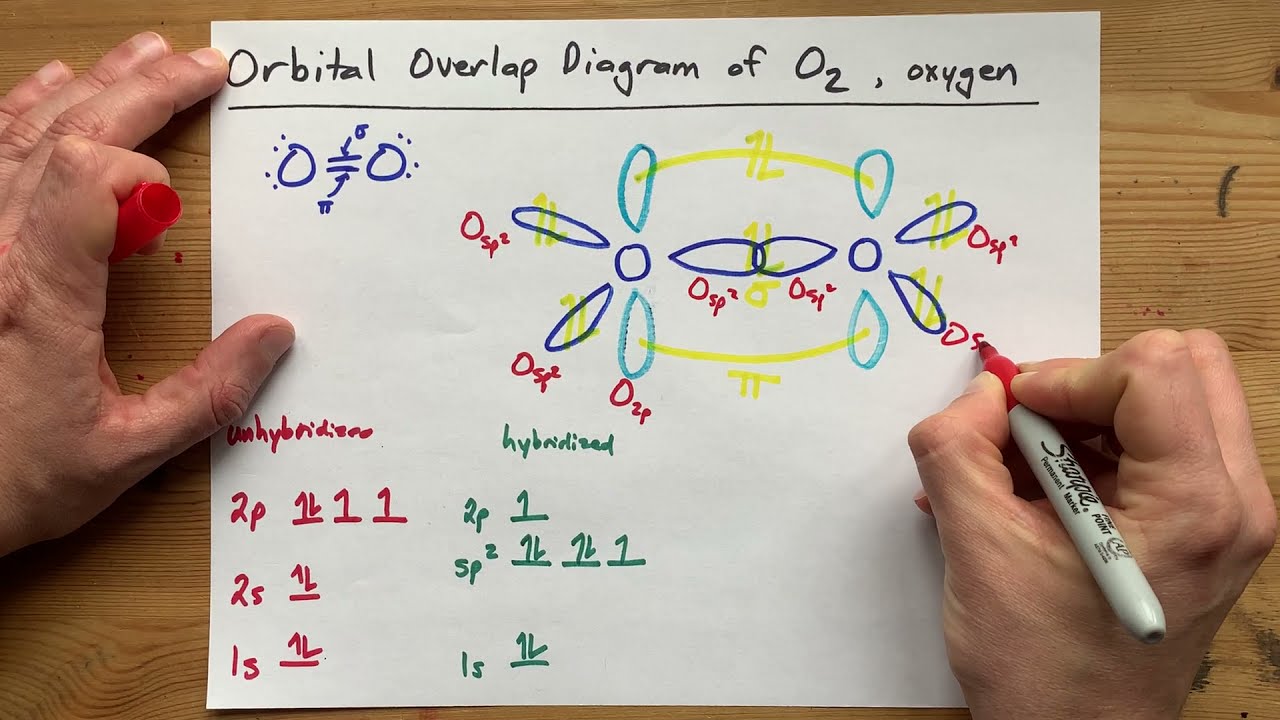
The hybridization of o3 (ozone) is sp2.Carbon atoms with hexagonal arrangements are bonded tightly in a honeycomb structure.In the ethane molecule, the bonding picture according to valence orbital theory is very similar to that of methane.
In this video, we use both of these methods to determine the hybridizations of atoms in various organic molecules.Add these two numbers together.
Last update images today Hybridization Of O3 Atoms
 Norris: No Need For Verstappen Apology After Crash
Norris: No Need For Verstappen Apology After Crash
The summer transfer window is open across Europe, and there is plenty of gossip swirling around. Transfer Talk brings you all the latest buzz on rumours, comings, goings and, of course, done deals!
TOP STORY: Man City eyeing move for Leipzig's OlmoManchester City are interested in a move for RB Leipzig forward Dani Olmo, according to Sky Sports Deutschland's Philipp Hinze.
A clause in the 26-year-old's contract that allows him to be signed for a fee of €60 million is reported to be active until the middle of July, but the Premier League club are yet to make "concrete contact" over a move for him despite the Spain international being keen on a switch to the Etihad Stadium.
Leipzig are said to be open to entering negotiations should his clause not be activated by the time it expires. Barcelona are one of the clubs also interested in a move for him, but their current financial situation means they could need help to meet any realistic demands.
Olmo contributed to nine goals in 21 Bundesliga games last season, and he has also starred in three matches at the European Championship while representing Spain.
PAPER GOSSIP
- Real Madrid currently lead the race for Lille centre-back Leny Yoro, reports Fabrizio Romano. It is reported that Liverpool and Paris Saint-Germain are interested in the 18-year-old, but his current preference is a switch to the Santiago Bernabeu. His representatives have informed the LaLiga club of their plan to avoid signing a new contract, as discussions continue between both sides over a transfer fee.
- Clubs in Saudi Arabia are looking at a potential move for Tottenham Hotspur and Brazil international forward Richarlison, reports Ben Jacobs. Al Ittihad and Al Hilal are said to have both discussed an approach for the 27-year-old, who could be allowed to leave the Premier League side for an offer of at least £60m.
- The decision to part ways with winger Federico Chiesa has been made by Juventus, reports Fabrizio Romano. Chiesa entered the final year of his contract recently, and it is reported that new manager Thiago Motta doesn't see him as a part of his future project with the Bianconeri. The Serie A club are reported to be preparing to sign a new winger once they offload the 26-year-old.
- Two clubs attempted to make a move for Bayern Munich centre-back Matthijs de Ligt so that he wouldn't join Manchester United, reports Bild's Christian Falk. Paris Saint-Germain and Liverpool are believed to have explored a move for the 24-year-old, but his current preference is to make the switch to Old Trafford. De Ligt made 16 starts in the Bundesliga last season.
- Manchester City left-back Sergio Gómez is close to completing a move to Real Sociedad, reports the Daily Mail. The 23-year-old is reportedly set to join the LaLiga side on a deal worth £8m, though the Citizens have included a clause that allows them the option to sign him back in the future.